Current Issue
Impact of a Multicomponent Nutraceutical on Cognitive Performance in Individuals with Mild Cognitive Impairment
Mauro Palumbo1*, Antonio Mainas2, Salvatore Travino3, Anna Papparella2,Francesco Scarcello2, Claudio Callipo2, Antonio Liguori2
1Department of Rehabilitation, Supra-District Complex Operational Unit, Local Health Authority BT (ASL BT), Italy
2General practitioner, Corigliano Rossano, 87064, Cosenza, Italy
3General practitioner, Cassano all’Ionio, 87011, Cosenza, Italy
*Corresponding author: Mauro Palumbo, Department of Rehabilitation, Supra-District Complex Operational Unit, Local Health Authority BT (ASL BT), Italy, E-mail: [email protected]
Received Date: March 02 2026
Publication Date: April 06, 2026
Citation: Palumbo M, et al. (2026). Impact of a Multicomponent Nutraceutical on Cognitive Performance in Individuals with Mild Cognitive Impairment. Nutraceutical Res. 5(1):22.
Copyright: Palumbo M, et al. © (2026).
ABSTRACT
Background: Mild to moderate mood disturbances are frequently observed in the general population and may negatively affect quality of life. In such cases, non-pharmacological approaches, including nutraceutical interventions, may represent a supportive strategy for mood regulation. This study aims to evaluate the effects of a nutraceutical formulation containing Griffonia simplicifolia, pomegranate, palmitoylethanolamide, vitamins B3, B6 and B12, and vitamin D on mood in subjects with mild to moderate mood disturbance not requiring antidepressant treatment. Methods: In this observational study, 200 adult subjects were enrolled in a primary care setting and followed for three months. Participants received one sachet per day of nutraceutical formulation. Mood was assessed at baseline (T0), after one month (T1), and after two months (T2) using the Zung Self-Rating Depression Scale. Treatment adherence, tolerability, and drop-out rates were also recorded. Results: A progressive reduction in mean Zung scale scores was observed, decreasing from 45.98 (±6.05) at T0 to 37.91 (±5.37) at T1 and 36.60 (±5.41) at T2. Treatment adherence was reported in 81% of patients at T1 and 93% at T2. Conclusions: Supplementation with the investigated nutraceutical was associated with an improvement in mood scores in subjects with mild to moderate mood disturbances over a three-month period.
Keywords: Mood Disturbances, Nutraceutical, Griffonia simplicifolia, Pomegranate, Palmitoylethanolamide, Vitamins B, Zung Self-Rating Scale
ABBREVIATION
PEA: Palmitoylethanolamide 5-HTTP: 5-hydroxytryptophan GP: General Practitioners
INTRODUCTION
Mood can be defined as a basic affective disposition that characterizes an individual's emotional experience, manifesting as both a relatively stable trait and a transient state susceptible to variation in response to internal and external stimuli. Mood represents a basic emotional state, generally of low intensity, that can persist over time and influence how an individual reacts to stimuli, even in the absence of a specific identifiable cause. Unlike emotions, mood is not linked to a specific event and can be subject to instability or dysregulation [1]. In fact, mood swings can occur, generally for the worse, without any apparent cause: the reason for this could be internal [2]. Mood, in fact, depends not only on external events and emotions that arise during a person’s life, but also on endogenous mechanisms and specific physiological processes, which can be altered in response to stressful conditions [3]. Specifically, mood is influenced by the presence and activity of certain neurotransmitters, such as dopamine, adrenaline, and serotonin, which are crucial neuromodulators for modulating the excitability of certain neurons [4]. Mood can have a direct impact on mental and physical health, including depression and anxiety, and on other conditions such as cardiovascular disease, addiction, psychological resistance, cognitive performance, aging, and longevity [5]. Overall, mental health is an essential component of overall well-being and exists in a continuum, like physical health [6]. In this context, various natural substances that have demonstrated a positive effect on mood may be beneficial for patients with mild to moderate mood disorders. Among plant-based substances, Griffonia simplicifolia is an evergreen plant in the Fabaceae family, native to central-western Africa. Its potential benefits for mood are mainly attributed to its 5-hydroxytryptophan (5-HTP) content, a metabolic intermediate in serotonin biosynthesis. Serotonin is a key neurotransmitter involved in mood regulation, and its deficiency has been associated with several emotional disorders [7]. Unlike serotonin, 5-HTP does not bind to plasma proteins and can cross the blood–brain barrier, with an estimated bioavailability of 60–70%, thereby increasing serotonin synthesis and release [7]. Pomegranate (Punica granatum) has also been suggested to exert beneficial effects on depressive symptoms, potentially due to its content of estrogenic compounds, such as estriol, estradiol, and estrone, which may modulate serotonergic activity [8]. Palmitoylethanolamide (PEA), an endogenous fatty acid amide with neuromodulatory properties, has been shown to increase attention and activity levels in animal models and to reduce mood disturbances in approximately 60% of patients [9]. In addition, vitamin D has been hypothesized to contribute to the maintenance of normal mood [10]. Human studies have demonstrated the presence of vitamin D receptors in several brain regions, and vitamin D metabolites appear to support neuronal integrity by upregulating neurotrophic factors, including nerve growth factor, neurotrophin-3, and neurotrophin-4, particularly in the hippocampus and neocortex [11]. Evidence also suggests that vitamin B6 (pyridoxine) plays a role in regulating symptoms associated with nervous hyperexcitability and mood instability. Deficiency of this vitamin has been associated with irritability, while restoration of adequate vitamin B6 levels appears to be associated with a rapid improvement in mood [12]. Vitamin B12 has likewise been implicated in mood regulation, with several studies reporting beneficial effects of vitamin B12 supplementation in patients with depressive symptoms [13]. Based on these observations, the present study aimed to evaluate the effect of a nutraceutical formulation containing Griffonia simplicifolia, pomegranate, palmitoylethanolamide (PEA), vitamins B3, B6 and B12, and vitamin D on mood in patients with mild to moderate alterations of normal mood who do not require treatment with antidepressant medications.
Materials and Methods
Study Design
This was an open interventional study aimed at evaluating the effect of a nutraceutical on mood in adults with mild to moderate mood disorders who were being treated by their general practitioner and were not eligible for treatment with antidepressant drugs. Patients were recruited and followed by General Practitioners (GP) in a primary care setting. After signing the informed consent form, eligible patients were instructed to take the nutraceutical at a dose of one sachet per day. Patients were observed for a period of three months, with 3 visits. The baseline visit was conducted at T0, followed by an intermediate visit 1 month after the start of treatment (T1) and a final visit two months later (T2). At each visit, the Zung scale was administered to assess mood. During the study, any adverse events and treatment discontinuations were also recorded, documenting their frequency and causes.
Inclusion and Exclusion Criteria
The study population included adults aged 18 or older, both sexes, in good general health, with mild to moderate mood disturbances that did not require antidepressant drug treatment. Patients with known intolerance to at least one of the product's components, individuals with serious medical or psychiatric conditions potentially responsible for mood changes, patients with a confirmed diagnosis of depression, those receiving antidepressant treatment or who had discontinued such treatment less than one month prior to the baseline visit, subjects taking other supplements or products related to complementary and alternative medicine for the management of mood disturbances, as well as pregnant or breastfeeding women, were excluded.
Treatment and Assessment
The nutraceutical was administered orally at a dose of 1 sachet per day throughout the study. During the baseline visit, in addition to completing the enrollment form and obtaining informed consent and privacy documentation, the Zung scale was administered, and the treatment was dispensed. At the mid-term visit, conducted 1 month after treatment initiation, the Zung scale was administered again, treatment compliance was assessed, the required packages for the subsequent 2 months were provided, and any adverse events occurring between T0 and T1 were recorded. At the final visit, performed three months after treatment initiation, the Zung scale was administered again, compliance was reassessed, and any adverse events occurring throughout the study period were documented. The Zung scale used in this study is a self-administered questionnaire consisting of 20 items designed to quantitatively assess depressive symptoms, encompassing both physiological and psychological dimensions. Responses are rated on a four-point scale ranging from “rarely” to “almost always.” A total score between 20 and 30 indicates no or minimal depression, scores between 31 and 43 indicate mild to moderate depression, values between 44 and 55 indicate moderate to severe depression, and scores between 56 and 80 indicate severe depression [14].
Primary and Secondary Endpoint
The primary endpoint of the study was to evaluate the effect of the nutraceutical on mood in patients with mild or moderate disorders who did not require antidepressant treatment. Secondary endpoints included evaluation of the nutraceutical's tolerability and analysis of dropouts, both in terms of frequency and causes.
Statistical Analysis
The sample size was calculated with a significance level of α = 0.05 and a statistical power of 1- β = 0.90. Based on the available literature, assuming an average difference of approximately 10 points between the scores at time T0 and T2, with a standard deviation of the difference equal to 20, a minimum sample size of 44 patients was estimated. The variables were initially described using descriptive analyses. For qualitative variables, absolute frequencies and percentages were reported, while for quantitative variables, the mean, median, quartiles, minimum and maximum values, and standard deviation were calculated. Changes in Zung scale scores at times T0, T1, and T2, being ordinal variables, were analyzed using the Friedman test. In cases of statistical significance, Dunn's post hoc tests were applied to identify differences between individual observation times. The complete ZUNG Scale questionnaire is presented in Table 1. For all analyses, the significance level was set at p < 0.05. Statistical analyses were performed using IBM SPSS Statistics software, version 28.
RESULTS
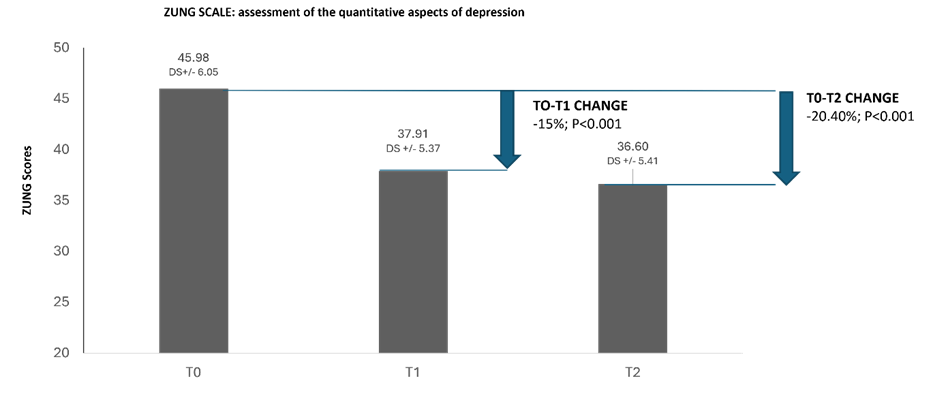
For this study 200 patients were enrolled and completed the study. As reported in Table 2, the study population consisted mainly of males (68.5%), while women accounted for 31.5% of the sample. The average age of participants was 53.79 years, with a standard deviation of ±15.02 years. Regarding the reported duration of mood swings, 10% reported a duration of 1 to 3 months, 8% reported 4 to 6 months, 8% reported 7 to 9 months, and 16% reported 10 to 12 months. Most subjects (59%) reported mood disorders lasting more than one year. The level of information patients perceived regarding symptoms associated with mood swings had an average score of 3.38, with a standard deviation of ±1.22. This value was measured on a five-point scale, where 0 corresponds to “not at all informed” and 4 to “very informed”. Figure 1 reports the average scores of the ZUNG Scale. The average scores on the Zung scale were recorded at the three observation times specified in the study. At baseline (T0), the average score was 45.98 with a standard deviation of ±6.05. After one month of treatment (T1), the average score was 37.91 with a standard deviation of ±5.37. At the final visit, three months after the start of treatment (T2), the average Zung scale score was 36.60 with a standard deviation of ±5.41.
Figure 1: Assessment of the quantitative aspects of depression with the ZUNG Scale.
A total score from 20 to 30 indicates no or low depression, from 31 to 43 indicates mild to moderate depression, from 44 to 55 indicates moderate to severe depression, from 56 to 80 indicates severe depression.
Table 1: Zung Scale questionnaire
|
|
Rarely |
Sometimes |
Often |
Almost always |
|
1. I feel more anxious and nervous than usual. |
||||
|
2. I feel afraid without any apparent reason. |
||||
|
3. I am easily frightened or experience panic. |
||||
|
4. I feel upset and as if I am about to break down. |
||||
|
5. I feel that everything is going well and that nothing bad will happen. |
||||
|
6. My arms and legs tremble. |
||||
|
7. I suffer from headaches and pain in my neck and back. |
||||
|
8. I feel weak and get tired easily. |
||||
|
9. I feel calm and can sit still easily. |
||||
|
10. I feel my heart beating fast. |
||||
|
11. I suffer from dizziness. |
||||
|
12. I feel like I might faint. |
||||
|
13. I can breathe easily. |
||||
|
14. I feel numbness or tingling in my fingers. |
||||
|
15. I suffer from stomach pain or indigestion. |
||||
|
16. I often feel the need to urinate. |
||||
|
17. My hands are usually dry and warm. |
||||
|
18. My face becomes easily flushed and red. |
||||
|
19. I fall asleep easily and wake up feeling rested. |
||||
|
20. I have nightmares. |
|
|
|
|
This test is used to assess the presence and possible severity of anxiety. Read the questionnaire carefully and use the definitions rarely, sometimes, often, almost always (corresponding to numerical values) to indicate the frequency of occurrence of the symptoms described in each sentence. Add up all the scores obtained and report the results to your doctor. Only he can correctly assess your psychophysical condition.
our doctor. Only he can correctly assess your psychophysical condition.
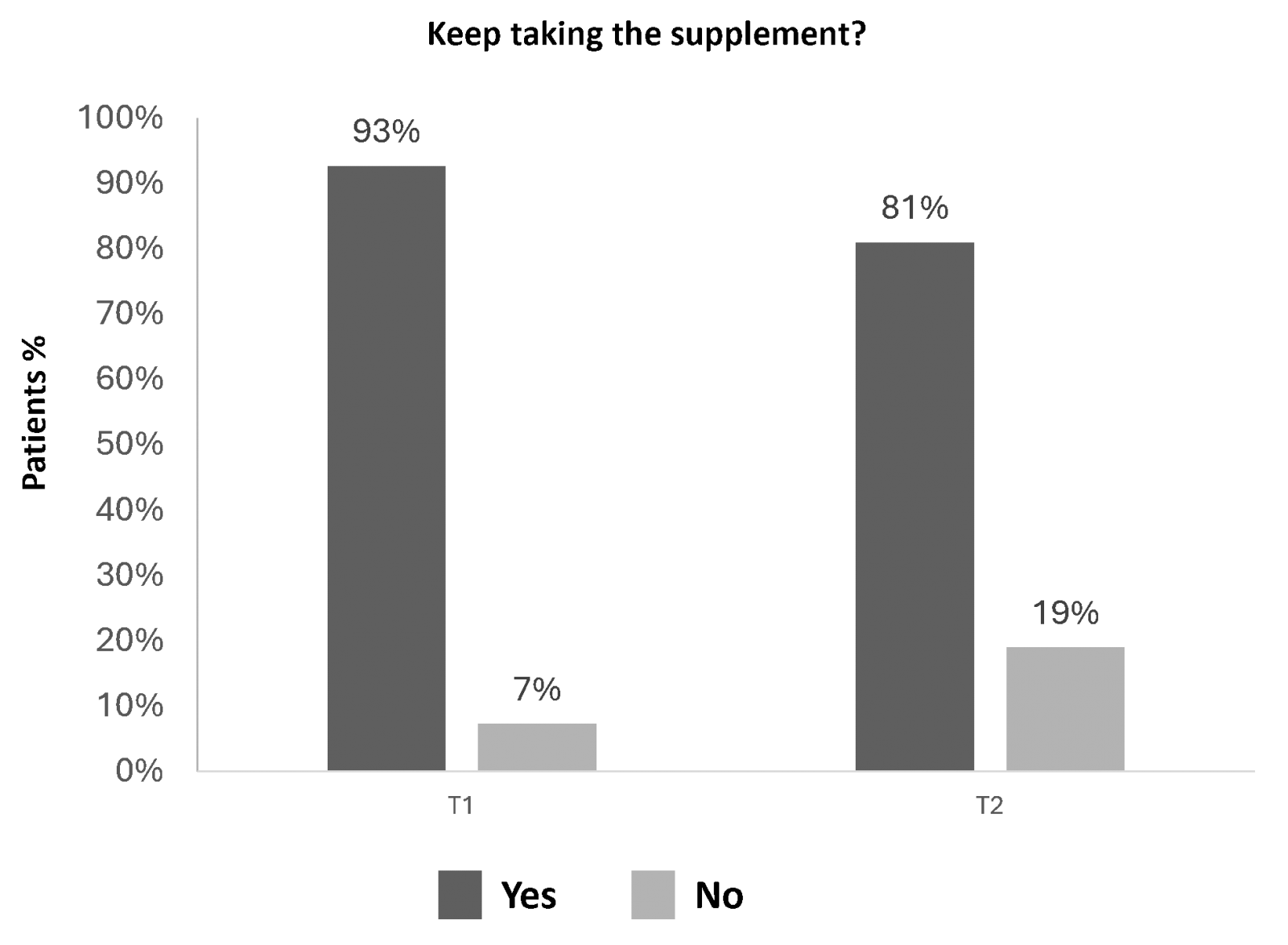
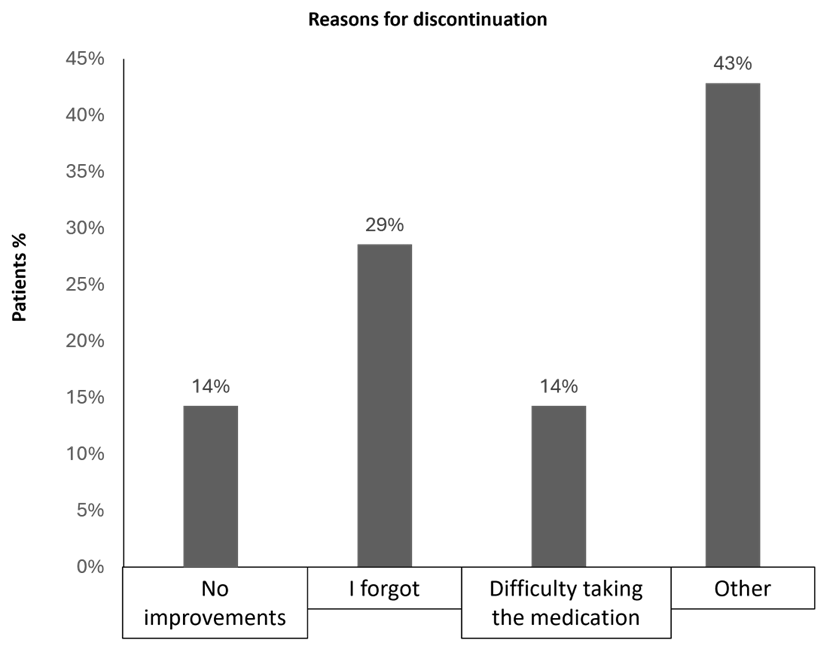
As shown in Figure 2, at T1, 93% of patients reported treatment adherence, while 7% reported non-adherence. At T2, 81% of patients were adherent to treatment, while 19% reported non-adherence. Figure 3 reports the reasons for discontinuation among the patients involved in the study. 29% of patients forgot the treatment, while 14% reported difficulty in taking the medication. The same percentage of patients reported no improvement as the major reason for discontinuation. As for the remaining patients, the reason for discontinuation was not specified (43%).
Figure 2: Assessment of treatment adherence among patient’s panel.
Figure 3: Reason for treatment discontinuation
Table 2: Patients’ demographics and characteristics
|
Characteristics |
% |
|
Male |
68.5 |
|
Female |
31.5 |
|
Mean age years (SD) |
53.79 (±15.02) |
|
How long have you been experiencing mood swings? |
|
|
· 1-3 months |
10 |
|
· 4-6 months |
8 |
|
· 7-9 months |
8 |
|
· 10-12 months |
16 |
|
· >1 year |
59 |
DISCUSSION
The results of this study show a reduction in average Zung scores during the observation period, with a decrease evident after the first month of treatment and maintained at the final assessment at three months. This trend is consistent with what has been reported in the literature for nutraceutical interventions aimed at supporting mood in individuals with mild or moderate disorders, in whom improvements in self-administered scale scores were observed as early as the first weeks of treatment [15-17]. The available evidence suggests that 5-hydroxytryptophan-based substances such as Griffonia simplicifolia, B vitamins, and vitamin D, may contribute to the regulation of the neurochemical mechanisms involved in mood [18-22], while compounds such as palmitoylethanolamide and pomegranate polyphenols have been associated with neuromodulatory effects and support for psychological well-being [23,24]. The data observed in this study are consistent with these biological hypotheses, although they do not allow causal relationships to be established. Another finding concerned treatment adherence, which remained high throughout the study, although a decreased at T2 compared to T1. The main reasons for discontinuation reported by patients were varied and included forgetfulness, difficulty in taking the treatment, and a perceived lack of benefit, aspects already described in observational studies conducted in the nutraceutical field and which reflect common critical issues in the management of non-pharmacological treatments.
Limitations of the Study
However, this study has some limitations that must be considered when interpreting the results. Firstly, the absence of a control group prevents us from ruling out a placebo effect or the influence of external factors on the observed improvement. Furthermore, the baseline clinical characterization of patients is limited, particularly regarding potentially relevant variables such as comorbidities, stress levels, sleep quality, or prior non-pharmacological treatments, which could have influenced mood patterns. Further limitations include the exclusive use of a self-administered scale for outcome assessment and the relatively short follow-up period, which precludes conclusions about the persistence of long-term effects. Future studies, preferably randomized and controlled, with greater population characterization and a longer follow-up period, will be necessary to confirm and further investigate the observations that have emerged.
CONCLUSIONS
Overall, the results of this study suggest that taking a nutraceutical containing Griffonia simplicifolia, pomegranate, palmitoylethanolamide, B vitamins, and vitamin D is associated with a reduction in Zung scale scores in patients with mild to moderate mood disorders who do not require antidepressant treatment. The improvement observed was evident after the first month of treatment and was maintained during the three-month follow-up. Further controlled, randomized studies will be necessary to confirm these findings and define more precisely the role of these interventions in clinical practice.
ACKNOWLEDGMENTS
All authors would like to thank Cristalfarma S.r.l. for providing the nutraceutical mix compound.
Author Contributions: All authors made substantial contributions to the conception and design of the study; acquisition, analysis, and interpretation of data. All authors participated in drafting the manuscript and critically revising it for important intellectual content. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work, ensuring the accuracy and integrity of any part of the study.
Funding: This research was funded by Cristalfarma S.r.l.Institutional Review Board Statement.
The study protocol was reviewed and approved by the appropriate Ethics Committee and was conducted in accordance with the international ethical standards as well as with the 1964 Helsinki Declaration, its later amendments, or comparable ethical standards.
Informed Consent Statement: Written informed consent was obtained from all participants prior to enrolment in the study.
Data Availability Statement: The data presented in this study are available upon request to the corresponding author.
Conflicts of Interest: The authors declare no conflicts of interest.
REFERENCES
- Rossi A, Di Stefano, R. (2024). Emotional instability: Terminological pitfalls and perspectives. Journal of Psychopathology. 30(1).
- Onda F. (2019). La Salute Mentale in Italia. Libro Bianco, Franco Angeli Editore: Milano, Italy.
- Marcolongo-Pereira C, Castro FCAQ, Barcelos RM, Chiepe KCMB, Rossoni Junior JV, Ambrosio RP, et al. (2022). Neurobiological mechanisms of mood disorders: Stress vulnerability and resilience. Frontiers in Behavioral Neuroscience. 16:1006836.
- Tsuboi D, Nagai T, Yoshimoto J, Kaibuchi K. (2024). Neuromodulator regulation and emotions: Insights from the crosstalk of cell signaling. Frontiers in Molecular Neuroscience. 17:1376762.
- Bar M. (2009). A cognitive neuroscience hypothesis of mood and depression. Trends in Cognitive Sciences. 13(11):456–463.
- Menefee DS, Ledoux T, Johnston CA. (2022). The importance of emotional regulation in mental health. American Journal of Lifestyle Medicine. 16(1):28-31.
- Muszyńska B, Łojewski M, Rojowski J, Opoka W, Sułkowska-Ziaja K. (2015). Natural products of relevance in the prevention and supportive treatment of depressionPsychiatria Polska. 49(3):435-453.
- Valdés-Sustaita B, López-Rubalcava C, González-Trujano ME, García-Viguera C, Estrada-Camarena E. (2017). Aqueous extract of pomegranate alone or in combination with citalopram produces antidepressant-like effects in an animal model of menopause: Participation of estrogen receptors. International Journal of Molecular Sciences. 18(12):2643.
- Sabelli H, Fink P, Fawcett J, Tom C. (1996). Sustained antidepressant effect of PEA replacement. The Journal of Neuropsychiatry and Clinical Neurosciences. 8(2):168-171.
- Annweiler C, Allali G, Allain P, Bridenbaugh S, Schott AM, Kressig RW, et al. (2009). Vitamin D and cognitive performance in adults: A systematic review. European Journal of Neurology. 16(10):1083-1089.
- Eyles DW, Smith S, Kinobe R, Hewison M, McGrath JJ. (2005). Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. Journal of Chemical Neuroanatomy. 29(1):21-30.
- Noah L, Dye L, Bois De Fer B, Mazur A, Pickering G, et al. (2021). Effect of magnesium and vitamin B6 supplementation on mental health and quality of life in stressed healthy adults: Post-hoc analysis of a randomised controlled trial. Stress Health. 37(5):1000-1009.
- Hintikka J, Tolmunen T, Tanskanen A, Viinamäki H. (2003). High vitamin B12 level and good treatment outcome may be associated in major depressive disorder. BMC Psychiatry. 3:17.
- Zung WW. (1965). A self-rating depression scale. Archives of General Psychiatry. 12:63–70.
- Sarris J, Byrne GJ, Stough C, Bousman C, Mischoulon D, Murphy J, et al. (2019). Nutraceuticals for major depressive disorder—More is not merrier: An 8-week double-blind, randomized, controlled trial. Journal of Affective Disorders. 245:1007-1015.
- Travica N, Teasdale S, Marx W. (2023). Nutraceuticals in mood disorders: Current knowledge and future directions. Current Opinion in Psychiatry. 36:54-59.
- Davis A, Pence J, Bloomer RJ. (2025). Nutraceuticals in the Treatment of Major Depressive Disorder. Nutraceuticals. 5(3):27.
- Javelle F, Lampit A, Bloch W, Häussermann P, Johnson SL, Zimmer P. (2020). Effects of 5-hydroxytryptophan on distinct types of depression: A systematic review and meta-analysis. Nutrition Reviews. 78:77–88.
- Nazhand A, Durazzo A, Lucarini M, Guerra F, Coêlho AG, Souto EB. (2024). Beneficial properties and sustainable use of a traditional medicinal plant: Griffonia simplicifolia. Challenges. 15(1):14.
- Kennedy DO. (2016). B vitamins and the brain: Mechanisms, dose and efficacy—A review. Nutrients. 8(2):68.
- AlGhamdi SA. (2024). Effectiveness of vitamin D on neurological and mental disorders. Diseases. 12(6):131.
- Skoczek-Rubińska A, Cisek-Woźniak A, Molska M, Heyser M, Trocholepsza M, Pietrzak S, et al. (2025). Impact of vitamin D status and supplementation on brain-derived neurotrophic factor and mood–cognitive outcomes: A structured narrative review. Nutrients. 17(16):2655.
- D'Aloia A, Molteni L, Gullo F, Bresciani E, Artusa V, Rizzi L, et al. (2021). Palmitoylethanolamide modulation of microglia activation: Characterization of mechanisms of action and implication for its neuroprotective effects. International Journal of Molecular Sciences. 22(6):3054.
- Estrada-Camerena E, López-Rubalcava C, Vega-Rivera NM, González-Trujano ME. (2024). Antidepressant- and anxiolytic-like effects of pomegranate: Is it acting by common or well-known mechanisms of action? Plants. 13(16):2205.
 Abstract
Abstract  PDF
PDF