Current Issue
Evaluation of the Efficacy and Safety of a Nutraceutical Containing Sodium Alginate, Sodium Bicarbonate, PEA, A Blend of Opuntia Ficus Indica, and Olea Europaea, Musa Paradisiaca, and Ginger in Patients with NERD
Carlo Calabrese1*, Nikolas Konstantine Dussias2, Eleonora Filippone2, Laura Melotti1,2, Enzo Spisni3, Francesco Bonomo4, Paolo Giochetti1,2, Fernando Rizzello1,2
1Department of Medical and Surgical Sciences, Alma Mater Studiorum University of Bologna, Bologna, Italy
2Department of Medical and Surgical Sciences, IBD Unit- IRCCS Azienda Ospedaliero-Universitaria- Policlinico Sant'Orsola-Malpighi, Bologna, Italy
3Department of Biological, Geological and Environmental Sciences, University of Bologna, 40126, Bologna, Italy.
4Cristalfarma Srl, 20143 Milan, Italy
*Corresponding author: Carlo Calabrese Department of Medical and Surgical Sciences, Alma Mater Studiorum University of Bologna, Via Zamboni, 33 - 40126, Bologna, Italy, Tel: +39 329 8195757, E-mail: [email protected]
Received Date: February 27, 2026
Publication Date: March 31, 2026
Citation: Calabrese C, et al. (2026). Evaluation of the Efficacy and Safety of a Nutraceutical Containing Sodium Alginate, Sodium Bicarbonate, PEA, A Blend of Opuntia Ficus Indica, and Olea Europaea, Musa Paradisiaca, and Ginger in Patients with NERD. Nutraceutical Res. 5(1):21.
Copyright: Calabrese C, et al. © (2026).
ABSTRACT
Background: Non-erosive reflux disease (NERD) is a common subtype of gastroesophageal reflux disease (GERD), frequently showing suboptimal response to proton pump inhibitors (PPIs). Eolo®, a nutraceutical formulation containing sodium alginate, sodium bicarbonate, palmitoylethanolamide (PEA), and herbal extracts, has been pro-posed as an adjunctive or alternative therapy to improve symptom control and mucosal protection. Methods: In this single-center, randomized, parallel-group study, 60 patients with NERD were assigned to: Eolo® plus half-dose PPIs for 12 weeks; half-dose PPIs alone for 12 weeks followed by Eolo®; or half-dose PPIs for 24 weeks. After 12 weeks, patients in groups A and B discontinued PPIs and continued Eolo® alone for an additional 12 weeks. Symptom severity and quality of life were evaluated using GERD-HRQOL and RSI questionnaires at baseline (T0), 12 weeks (T1), and 24 weeks (T2). Results: Eolo®-treated groups showed non-inferiority to PPIs alone in improving GERD-HRQOL and RSI scores, with notable reductions in regurgitation and heartburn. No significant differences between groups were observed. The nutraceutical demonstrated a favorable safety profile, with mild, transient adverse events. Compliance exceeded 98%, and 93% of participants reported good palatability. Conclusion: Eolo®, as adjunctive or standalone therapy, provides symptom relief comparable to PPIs in NERD patients and may offer additional mucosal protection. Further studies are needed to confirm long-term benefits.
KEYWORDS: Gastroesophageal Reflux Disease, Non-Erosive Reflux Disease, Musa paradisiaca, Olea europaea, Opuntia ficus-indica, Zingiber officinale, Palmitoylethanolamide
ABBREVIATIONS
NERD: Non-erosive Reflux Disease
GERD: Gastroesophageal Reflux Disease
PPIs: Proton Pump Inhibitors
PEA: Palmitoylethanolamide
TLESRs: Transient Lower Esophageal Sphincter Relaxations
RDQ: Reflux Disease Questionnaire
INTRODUCTION
Gastroesophageal reflux disease (GERD) is a widespread condition in Western countries, with heartburn, its primary symptom, affecting up to 26% of the population weekly. Although the prevalence of GERD varies geographically, it is steadily increasing worldwide. Over the past decade, it has become evident that GERD manifests in two distinct phenotypes. Some patients exhibit esophageal mucosal lesions, such as erosive esophagitis, while the majority (up to 70%) have normal mucosa observed during endoscopy. This latter group is classified as having non-erosive reflux disease (NERD) [1]. Symptomatically, alongside classic signs like heartburn and regurgitation, many patients experience a reduced quality of life due to disrupted sleep, physical discomfort, anxiety, and dissatisfaction with their sexual health. While reflux episodes can occur throughout the day, nocturnal symptoms significantly impact quality of life and are linked to disease complications [2]. Proton pump inhibitors (PPIs) remain the first-line treatment for GERD, achieving an 80–85% healing rate for esophageal lesions, including ulcers, and reducing the risk of complications. Studies indicate symptom relief in 56–76% of cases, though patients with NERD experience less benefit. A widely cited systematic review reported that symptom relief in NERD patients is approximately 20% lower compared to those with erosive esophagitis. Moreover, a large survey conducted by the American Gastroenterological Association (AGA) revealed that over 55% of individuals with GERD symptoms, despite PPI treatment, continue to experience disruptions to their quality of life, encompassing both erosive and non-erosive cases [3]. Some studies have shown that both acidic and non-acidic reflux can induce histopathological changes, which have been clearly documented by electron and light microscopy in most NERD patients [4,5]. Specifically, the dilation of intercellular spaces between adjacent cells in the esophageal epithelium has become a hallmark of microscopic esophagitis. This intercellular gap increases permeability, allowing hydrogen ions, pepsin, bile, and other substances to penetrate the esophageal submucosa. These substances then reach nerve fibers, whose stimulation leads to the typical symptom of heartburn. Several studies have suggested a synergistic effect between acid and duodenogastric reflux in causing these lesions [6-8]. The role of pepsin in the pathogenesis of extra-esophageal manifestations of GERD is also gaining in-creasing recognition [9]. An ideal therapy for NERD patients should not only target acid secretion but also address the pathophysiological features. Specifically, it should provide a barrier to, and/or bind, the remaining aggressive components of the refluxate (such as weakly acidic content and pepsin). To achieve these goals, a specifically de-signed and developed dietary supplement, Eolo®, was created. It consists of a blend of natural substances recognized by the Ministry of Health, forming a macromolecular complex that counteracts the harmful components of reflux. The blend, taken at a dosage of 2 sticks/day, is composed of sodium alginate, sodium bicarbonate, PEA WD, a blend of Opuntia ficus-indica and Olea europaea, Musa paradisiaca, and ginger extract (20% gingerols). The components of the nutraceutical Eolo® are well-known physiological substances. A blend of two herbal extracts, Opuntia ficus-indica and Olea europaea, was designed to support and maintain mucosal health and may be useful in gastric disorders. Opuntia ficus-indica extract has shown mucoprotective and anti-inflammatory activity in vitro [10], while Olea europaea extract has demonstrated anti-ulcer properties [11,12] as well as antioxidant and mucoprotective activity [13]. Palmitoylethanolamide (PEA) is a long-chain fatty acid amide that combines palmitic acid with an ethanolamine group. PEA is naturally produced by the body, but its synthesis decreases with age. It is an endocannabinoid-like compound, meaning that while it does not bind directly to cannabinoid receptors, it modulates their function. PEA participates in the biological response to endogenous cannabinoids, prevents their degradation, regulates inflammation, and influences pain perception. Regarding gastroesophageal reflux, a 2009 study demonstrated that delta-9-THC significantly reduced esophageal sphincter pressure, confirming previous findings in dogs and suggesting that cannabinoid receptors play a role in transient lower esophageal sphincter relaxations (TLESRs) in humans [14]. A study first identified CB1 receptors in the human esophageal epithelium [15]. Additionally, CB2 receptors, which are mainly found in immune cells along the gastrointestinal tract, have also been de-scribed in the enteric nervous system [16]. These findings support the hypothesis that PEA may have a beneficial effect in GERD through interactions with the endocannabinoid system of the gastrointestinal tract [17]. Musa paradisiaca (commonly known as dwarf banana) is recognized by the Italian Ministry of Health for its gastric acidity regulation and digestive support [18]. As early as 1987, studies suggested that banana extracts enhance gastric mucosal resistance and promote ulcer healing [19]. The protective effects of banana have been further supported by its role in increasing gastric defensive factors, including mucin secretion, mucosal glycoproteins, increased cell proliferation, and regulation of acid-pepsin secretion [20–22]. Ginger (Zingiber officinale) is another plant recognized by the Italian Ministry of Health for its digestive and anti-nausea properties [23]. A 2013 review highlighted ginger’s free radical scavenging and lipid peroxidation inhibition activities, which contribute to its gastroprotective effects [23]. Ginger promotes gastric motility and enhances gastric emptying, which is particularly relevant for GERD patients. The prokinetic effects of ginger have been linked to improved gastric motility and reduced dyspeptic symptoms, including stimulation of gastric antral contractions in patients with functional dyspepsia [24]. Since prokinetic agents are often included in GERD management to facilitate esophageal and gastric emptying, ginger may offer similar benefits, reducing postprandial reflux episodes. Clinical trials indicate that sodium alginate is effective in managing GERD symptoms, particularly heartburn and acid regurgitation. A randomized clinical trial [25] demonstrated that sodium alginate, either alone or combined with other agents such as antacids, significantly reduced GERD symptoms. The addition of sodium bicarbonate helps neutralize stomach acid, though its effect is temporary. However, it provides rapid symptom relief, making it a useful rescue therapy for breakthrough re-flux symptoms. Combination products containing sodium alginate and sodium bicarbonate have been shown to effectively control GERD symptoms with a low risk of side effects. Studies comparing alginate-antacid formulations with PPIs suggest that they offer similar symptomatic relief, particularly in mild GERD cases. An experimental in vitro study on an esophageal mucosal cell line demonstrated that each of these com-pounds reduced inflammation induced by hyperacidity. The CP-B cell line (ATCC, CP-52731-CRL-4028) was grown in a 24-well plate and exposed to acidic conditions (pH 4.5) for 15 minutes. Under these conditions, cells exhibited inflammatory responses, including the secretion of pro-inflammatory cytokines, which were measurable in the culture medium. Pre-treatment with the four nutraceutical compounds prior to acid exposure demonstrated protective effects, mitigating the inflammatory response [26]. In this prospective trial, the efficacy and safety of Eolo®, in combination with acid suppression therapy or alone, was compared to acid suppression therapy in patients with non-erosive reflux disease (NERD).
MATERIALS AND METHODS
Study Design
This single-center, randomized study employed a parallel-group design. It was conducted in compliance with the International Conference on Harmonization of Technical Requirements for the Registration of Pharmaceutical Products for Human Use (ICH), the Guidelines for Good Clinical Practice (GCP), and the Declaration of Helsinki (1996 version, amended in October 2000). Ethical approval was obtained from the IRCCS Bologna Ethics Committee (approval number 3/2023/Sper/AOUBo). All eligible participants provided written informed consent before their inclusion in the study.
Inclusion and Exclusion Criteria
Eligible participants were aged between 18 and 80 years and presented with typical reflux symptoms persisting for at least three months, occurring at least three times per week during the month preceding the screening visit. All patients had been receiving standard-dose proton pump inhibitor (PPI) therapy for a minimum of six weeks prior to enrollment (pantoprazole 40 mg, esomeprazole 40 mg, lansoprazole 30 mg, omeprazole 20 mg, or rabeprazole 20 mg). The diagnosis of NERD was confirmed by the absence of macroscopic lesions of the distal esophageal mucosa on upper gastrointestinal endoscopy performed within six months before screening, along with a positive Reflux Disease Questionnaire (RDQ) score ≥ 8. Patients with erosive esophagitis, Barrett’s esophagus, gastric or duodenal ulcer, history of major gastrointestinal surgery, atopy or food intolerances, thyroid disease, diabetes mellitus, metabolic syndrome, or those who were pregnant or breastfeeding were excluded from the study.
Study Procedures
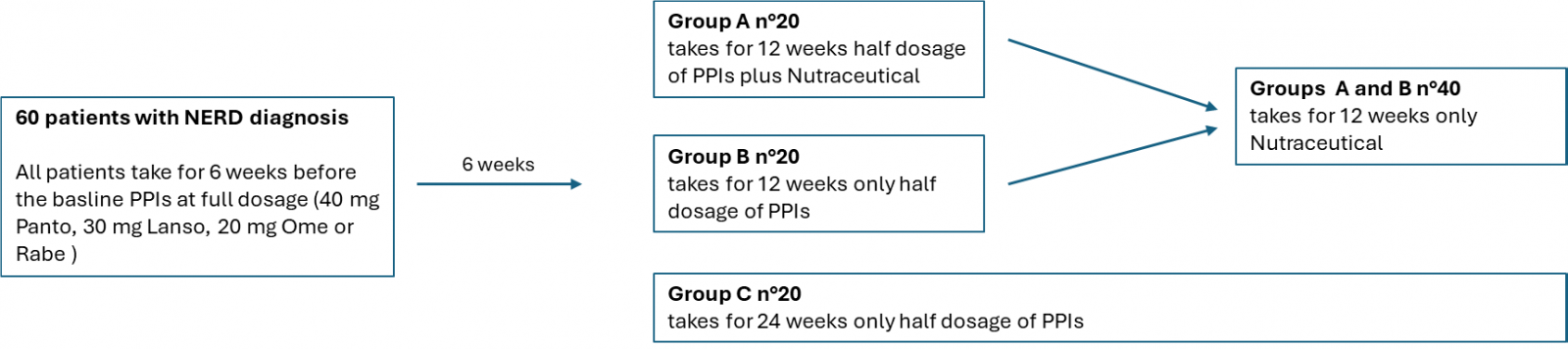
Figure 1 reports the study flowchart of patients’ allocations and treatment phases. During a 6-week run-in period, all participants received a full-dose PPI regimen. At baseline (Visit 1), patients were randomized using a computer-generated sequence into three groups: Group A received half-dose PPIs in combination with the nutraceutical formulation for 12 weeks; Group B received half-dose PPIs alone for 12 weeks; and Group C (control group) received half-dose PPIs for 24 weeks. After the initial 12-week treatment phase, patients in Groups A and B discontinued PPIs and continued treatment with the nutraceutical alone for an additional 12 weeks to evaluate its potential role in maintaining symptom control and quality of life in the absence of ac-id-suppressive therapy. The nutraceutical was administered at a dosage of two sticks per day, one taken one hour after breakfast and the other at bedtime. Treatment effectiveness was assessed at baseline (T0), after 12 weeks (T1), and at the end of the study (T2) using patient diaries and validated questionnaires, including the Gastroesophageal-geal Reflux Disease–Health-Related Quality of Life (GERD-HRQOL) questionnaire and the Reflux Symptom Index (RSI), to evaluate symptom severity and disease-related quality of life.
Figure 1: Study flowchart of patient allocation and treatment phases.
Safety and Compliance Assessments
All adverse events (AEs), defined as any unintended or unfavorable symptoms or signs potentially related to the study drugs, were recorded. Safety and tolerability as-assessments were performed throughout the study. The palatability of the nutraceutical product was evaluated weekly using a 4-point scale: Excellent, Good, Irrelevant, and Bad. Patient compliance was determined as the percentage of the product consumed, based on the count of returned products at Visits 2 and 3. A compliance rate of 80–100% was considered acceptable.
Statistical Methods
Descriptive statistics were calculated for all variables. For qualitative variables, the number and percentages were reported. For quantitative variables, the main indices (mean and standard deviation) were provided. Descriptive statistics were calculated separately for each group. For each item, delta variables were calculated as the difference between: T2-T0, T1-T0, and T2-T1. For variables measured at T0, T1, and T2, the Friedman test was used to assess whether their distribution across the three time points differed significantly. If significance was found, Dunn’s post hoc tests were performed to identify specific differences. A significant level of p < 0.05 was applied to all statistical tests. All analyses were conducted using IBM SPSS Statistics v.28 software.
RESULTS
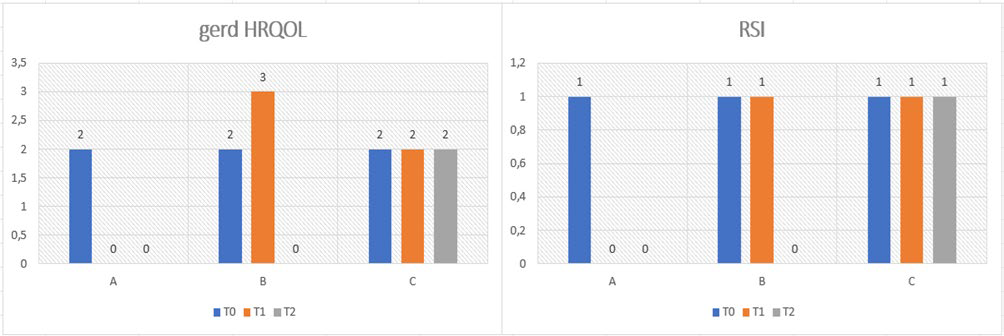
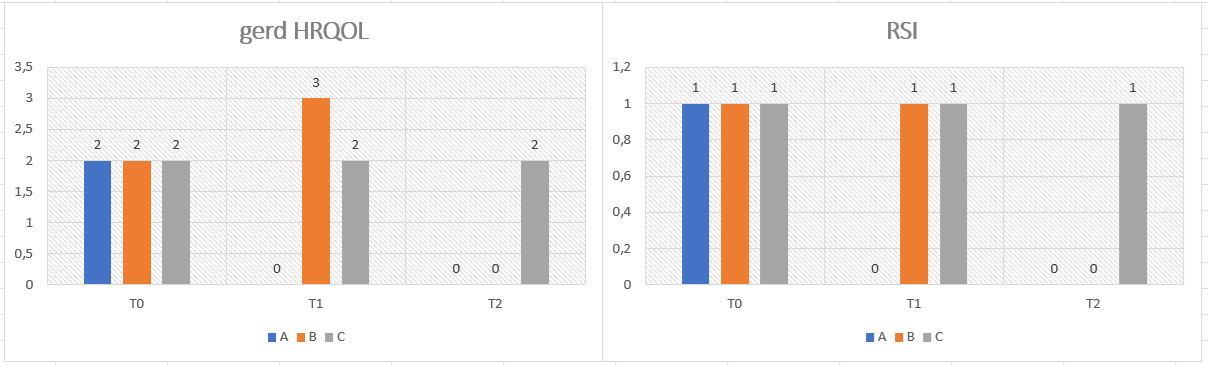
General characteristics are presented in Table 1, which summarizes the baseline demographics and clinical characteristics of the study groups. No significant differences were observed among the groups. Regarding the primary endpoint, Figure 2 illustrates the response to the GERD-HRQOL and RSI questionnaires for each group. Notably, Group A, during the first follow-up period - when patients took PPIs at half dosage in addition to the nutraceutical - maintained the same quality of life (QoL) as during the run-in period (full-dose PPIs). This effect persisted throughout the second follow-up period, during which patients took only the nutraceutical. The results obtained by Group B, which received only half-dose PPIs in the first follow-up and then switched to nutraceutical monotherapy in the second follow-up, demonstrated non-inferiority to the gold standard (PPIs). This was evident both during the half-dose treatment phase and the run-in period. All assessed symptoms improved with both treatments; however, the reduction in regurgitation was more pronounced with the nutraceutical alone. The intergroup analysis (Figure 3) presents the statistical comparison between the three groups at each follow-up period after the run-in (T0-T1 and T1-T2). Patients in all groups (A, B, and C) experienced comparable improvements in GERD-specific quality of life and Reflux Symptom Index (RSI) scores, with no significant differences between groups. The therapeutic gain associated with the nutraceutical for regurgitation incidence was 28.5% in Group A and 12.3% in Group B, compared to Group C. Additionally, Table 2 shows a slight trend towards improvement in items 5, 6, and 7 of the GERD-HRQOL questionnaire when the nutraceutical was taken either as an add-on to PPIs or as monotherapy. Both treatments (Groups A and B) were well tolerated, with mild adverse events (AEs) reported in similar proportions across the groups. No serious AEs or treatment-related discontinuations were observed. The most reported AEs included mild gastrointestinal discomfort (3%) and transient bloating (2%), both of which were resolved spontaneously. Treatment compliance exceeded 98% in both groups (A and B), indicating strong adherence to the prescribed regimens. Patient feedback on the palatability of the nutraceutical was overwhelmingly positive, with 93% of participants rating the product as “excellent” or “good”. Although not a primary endpoint, the study also suggested a potential mucoprotective effect of the nutraceutical. This was inferred from the reduction in self-reported regurgitation and burning sensation, indicating a possible beneficial impact on esophageal mucosal integrity.
Figure 2: Results of GERD-HRQOL and RSI questionnaire as an average at each follow-up.
Figure 3: Results as ratio between the three groups (A, B and C).
Table 1: Baseline characteristics of NERD patients.
|
|
Group A |
Group B |
Group C |
P value |
|
Sex (M/F) |
11/9 |
10/10 |
11/9 |
NS |
|
Age (mean +/- SD) |
60.6 (12.8) |
59.6 +/-14.9 |
59.3 +/-11.5 |
NS |
|
Range ((min, max) |
31-79 |
30-78 |
49-77 |
NS |
|
BMI (kg/m2) |
23.8+/-3.1 |
23.3 +/-3.3 |
22.9 +/-3 |
NS |
|
GERD Total symptoms score |
7.3+/-2.4 |
7.19+/- 2.6 |
7.21+/- 2.7 |
NS |
|
Proportion of patients with >3 GERD symptoms (%) |
45.1 |
44.9 |
44.8 |
NS |
|
Heartburn |
84.1 |
83.2 |
85.4 |
NS |
|
Retrosternal pain |
59.1 |
58.4 |
59.2 |
NS |
|
Acid regurgitation |
70.2 |
74.1 |
72.3 |
NS |
|
Acid total in the mouth |
23.5 |
24.1 |
21.2 |
NS |
|
PPI Treatment |
||||
|
Omeprazole |
2 |
1 |
2 |
|
|
Lansoprazole |
2 |
2 |
3 |
|
|
Rabeprazole |
2 |
2 |
1 |
|
|
Pantoprazole |
7 |
9 |
8 |
|
|
Esomeprazole |
7 |
6 |
6 |
|
|
GERD-HRQOL group A |
|||
|
ITEM 5 |
ITEM 6 |
ITEM 7 |
|
|
T0 |
0.9 |
0.6 |
0.6 |
|
T1 |
0.0 |
0.2 |
0.0 |
|
T2 |
0.0 |
0.1 |
0.0 |
|
GERD-HRQOL group B |
|||
|
ITEM 5 |
ITEM 6 |
ITEM 7 |
|
|
T0 |
0.9 |
0.6 |
0.6 |
|
T1 |
1.15 |
0.6 |
0.6 |
|
T2 |
0.0 |
0.0 |
0.0 |
|
GERD-HRQOL group C |
|||
|
ITEM 5 |
ITEM 6 |
ITEM 7 |
|
|
T0 |
0.9 |
0.6 |
0.6 |
|
T1 |
0.9 |
0.6 |
0.6 |
|
T2 |
0.9 |
0.6 |
0.6 |
Table 2: Results as an average at each follow-up for items 5,6,7 of GERD-HRQOL questionnaire.
DISCUSSION
The findings of this study demonstrate that Eolo® is a viable therapeutic option for managing NERD, showing non-inferiority compared to standard PPI therapy. Patients who received Eolo® alongside a reduced PPI dose (Group A) experienced symptom relief comparable to those on PPIs alone, while patients who transitioned to Eolo® monotherapy (Group B) maintained symptom control without deterioration. These results suggest that Eolo® may function both as an effective adjunct to PPIs and as an independent treatment strategy for NERD, addressing not only acid suppression but also additional pathophysiological mechanisms underlying the disease. A key observation is that Eolo® was not less effective than standard PPI therapy in controlling GERD-related symptoms. The GERD-HRQOL and RSI questionnaire confirmed that both treatment groups (A and B) experienced significant symptom improvements, with no statistically significant differences between them. Notably, symptom relief persisted even after PPI discontinuation, as observed in the second phase of the study (T1-T2), where patients who transitioned to Eolo® alone maintained comparable quality-of-life scores. The non-inferiority of Eolo® is particularly relevant given the limitations of PPI therapy in NERD patients. PPIs primarily target acid suppression but fail to address other reflux components, such as weakly acidic reflux and pepsin-related mucosal damage. In contrast, Eolo® incorporates a macromolecular complex of natural compounds designed to create a protective barrier, neutralize acidity, and reduce esophageal permeability. This mechanism of action may explain why patients in the Eolo® groups reported a greater reduction in regurgitation compared to those in the PPI-only group. Since regurgitation is often more resistant to PPIs than heartburn, this finding highlights a potential advantage of Eolo® in addressing persistent GERD symptoms. This study also demonstrated that Eolo® is well-tolerated, with a safety profile comparable to PPIs. Across all treatment groups, adverse events (AEs) were mild and transient, primarily consisting of minor gastrointestinal discomfort (3%) and transient bloating (2%). No serious AEs or treatment-related discontinuations were reported, reinforcing Eolo®'s safety as a therapeutic option. From a long-term management perspective, the safety of Eolo® is particularly relevant. Chronic PPI use has been associated with concerns such as increased risks of osteoporosis, kidney disease, and gut microbiota alterations. Although the clinical significance of these risks remains debated, the ability to effectively manage NERD symptoms with a natural, well-tolerated alternative could benefit patients who either do not respond adequately to PPIs or prefer to minimize pharmaceutical dependency. Patient adherence is critical in managing chronic conditions such as NERD, where symptom recurrence is common upon treatment discontinuation. Treatment compliance in the Eolo® groups exceeded 98%, reflecting strong adherence to the prescribed regimen. A key factor contributing to this high adherence was the positive palatability of Eolo®, with 93% of patients rating it as either "excellent" or "good." Given that taste and ease of administration significantly influence long-term compliance, Eolo®’s favorable sensory characteristics may offer a practical advantage over other treatments, particularly for patients with medication aversion or difficulty swallowing pills. The results of this study suggest that Eolo® may serve as an effective alternative to standard acid suppression therapy for NERD, particularly in patients who experience partial symptom relief with PPIs or seek a more holistic approach to disease management. Eolo®’s combination of ac-id-neutralizing, mucoprotective, and anti-inflammatory properties aligns well with the complex pathophysiology of NERD, addressing not only acid reflux but also esophageal hypersensitivity and mucosal barrier dysfunction. In summary, the findings of this study support the non-inferiority of Eolo® compared to PPI therapy in managing NERD symptoms, with comparable efficacy, excellent safety, and high patient adherence. Given its favorable tolerability and palatability, Eolo® represents a promising therapeutic option that could potentially reduce reliance on long-term PPI use while maintaining effective symptom control. Further research is warranted to explore the long-term benefits of Eolo®, particularly its role in mucosal healing and prevention of disease progression. Future studies should also investigate whether Eolo® can be effectively integrated into broader GERD management strategies, such as life-style modifications and dietary interventions, to enhance patient outcomes.
Limitations of the Study
This study has several limitations that should be acknowledged. First, the open-label design and the absence of a placebo-controlled arm may have introduced expectation bias, particularly given that patient-reported outcomes (GERD-HRQOL and RSI) were used as primary endpoints. Second, although symptom improvement in the nutraceutical groups was comparable to that observed with PPIs, the study was not designed as a formal non-inferiority trial, as no predefined non-inferiority margin or sample size calculation was established. Third, the objective reflux parameters, such as esophageal acid exposure time or impedance-based metrics, were not assessed; there-fore, the suggested mucoprotective effect of the nutraceutical remains indirect and requires confirmation through physiological measurements. Finally, the single-center design and relatively small sample size may further limit the generalizability of the findings. Future randomized, double-blind, placebo-controlled studies including impedance-pH monitoring are warranted to better define the role of this nutraceutical approach in the management of NERD.
Conflicts of Interest: The authors declare no conflicts of interest.
REFERENCES
- Yamasaki T, Hemond C, Eisa M, Ganocy S, Fass R. (2018). The changing epidemiology of gastroesophageal reflux disease: Are patients getting younger? J Neurogastroenterol Motil. 24(4):559-569.
- Young A, Kumar MA, Thota PN. (2020). GERD: A practical approach. Cleveland Clinic Journal of Medicine. 87(4):223-230.
- Gyawali CP, Fass R. (2018). Management of gastroesophageal reflux disease. Gastroenterology. 154(2):302-318.
- Haggitt RC. (2000). Histopathology of reflux-induced esophageal and supraesophageal injuries. The American journal of medicine.. 108(4):109S-111S.
- Rizza L, Frasca G, Nicholls M, Puglia C, Cardile V. (2012). Caco-2 cell line as a model to evaluate mucoprotective properties. International Journal of Pharmaceutics. 422(1–2):318-322.
- Sharma P, Yadlapati R. (2021). Pathophysiology and treatment options for gastroesophageal reflux disease: Looking beyond acid. Annals of the New York Academy of Sciences.1486(1):3-14.
- Vaezi MF, Singh S, Richter JE. (1995). Role of acid and duodenogastric reflux in esophageal mucosal injury: A review of animal and human studies. Gastroenterology. 108(6):1897-1907.
- Calabrese C, Bortolotti M, Fabbri A, Areni A, Cenacchi G, Scialpi C, et al. (2005). Reversibility of GERD ultrastructural alterations and relief of symptoms after omeprazole treatment. The American journal of gastroenterology. 100(3):537-542.
- Durazzo M, Lupi G, Cicerchia F, Ferro A, Barutta F, Beccuti G, et al. (2020). Extra-esophageal presentation of gastroesophageal reflux disease: 2020 update. Journal of Clinical Medicine. 9(8):2559.
- Galati EM, Monforte MT, Tripodo MM, d’Aquino A, Mondello MR. (2001). Antiulcer activity of Opuntia ficus indica (L.) Mill. (Cactaceae): Ultrastructural study. Journal of Ethnopharmacology.76(1):1-9.
- Galati EM, Pergolizzi S, Miceli N, Monforte MT, Tripodo MM. (2002). Study on the increment of the production of gastric mucus in rats treated with Opuntia ficus indica (L.) Mill. cladodes. Journal of Ethnopharmacology. 83(3):229-233.
- Dekanski D, Janićijević-Hudomal S, Ristić S, Radonjić NV, Petronijević ND, Piperski V, et al. (2009). Attenuation of cold restraint stress-induced gastric lesions by an olive leaf extract. General Physiology and Biophysics. 28(2):135-142.
- Alecci U, Bonina F, Bonina A, Rizza L, Inferrera S, Mannucci C, et al. (2016). Efficacy and safety of a natural remedy for the treatment of gastroesophageal reflux: A double-blinded randomized-controlled study. Evidence-based Complementary and Alternative Medicine. 2016:2581461.
- Beaumont H, Jensen J, Carlsson A, Ruth M, Lehmann A, Boeckxstaens G. (2009). Effect of delta9-tetrahydrocannabinol, a cannabinoid receptor agonist, on the triggering of transient lower oesophageal sphincter relaxations in dogs and humans. British Journal of Pharmacology. 156(1):153-162.
- Calabrese C, Spisni E, Liguori G, Lazzarini G, Valerii MC, Strillacci A, et al. (2010). Potential role of the cannabinoid receptor CB in the pathogenesis of erosive and non-erosive gastro-oesophageal reflux disease. Alimentary Pharmacology & Therapeutics. 32(4):603-611.
- Wright K, Rooney N, Feeney M, Tate J, Robertson D, Welham M et al. (2005). Differential expression of cannabinoid receptors in the human colon: Cannabinoids promote epithelial wound healing. Gastroenterology. 129(2):437-453.
- Spigarelli R, Calabrese C, Spisni E, Vinciguerra S, Saracino IM, Dussias NK, et al. (2024). Palmitoylethanolamide (PEA) for prevention of gastroesophageal inflammation: Insights from in vitro models. Life. 14(9):1221.
- Direzione Generale per l’Igiene e la Sicurezza degli Alimenti e la Nutrizione. (2019). Decreto 26 luglio 2019.
- Mukhopadhyaya K, Bhattacharya D, Chakraborty A, Goel RK, Sanyal AK. (1987). Effect of banana powder (Musa sapientum var. paradisiaca) on gastric mucosal shedding. Journal of Ethnopharmacology. 21(1):11-19.
- Sanyal AK, Banerjee CR, Das PK. (1965). Studies on peptic ulceration. Part II. Role of banana in restraint- and prednisolone-induced ulcer in albino rats. Archives internationales de pharmacodynamie et de thérapie. 155:244-288.
- Goel RK, Gupta S, Shankar R, Sanyal AK. (1986). Antiulcerogenic effect of banana powder (Musa sapientum var. paradisiaca) and its effect on mucosal resistance. Journal of Ethnopharmacology. 18(1):33-44.
- Gael RK, Govinda Das D, Sanyal AK. (1985). Effect of vegetable banana powder on changes induced by ulcerogenic agents on dissolved mucosubstances in gastric juice. Indian Journal of Gastroenterology. 4(4):249-251.
- Haniadka R, Saldanha E, Sunita V, Palatty PL, Fayad R, Baliga MS. (2013). A review of the gastroprotective effects of ginger (Zingiber officinale Roscoe). Food & Function. 4(6):845-855.
- Hu ML, Rayner CK, Wu KL, Chuah SK, Tai WC, Chou YP, et al. (2011). Effect of ginger on gastric motility and symptoms of functional dyspepsia. World Journal of Gastroenterology. 17(1):105-110.
- Reimer C, Lødrup AB, Smith G, Wilkinson J, Bytzer P. (2016). Randomised clinical trial: Alginate (Gaviscon Advance) vs. placebo as add-on therapy in reflux patients with inadequate response to a once daily proton pump inhibitor. Alimentary Pharmacology & Therapeutics. 43(8):899-909.
- Kulich KR, Calabrese C, Pacini F, Vigneri S, Carlsson J, Wiklund IK. (2004). Psychometric validation of the Italian translation of the gastrointestinal symptom-rating scale and quality of life in reflux and dyspepsia questionnaire in patients with gastro-oesophageal reflux disease. Clinical drug investigation. 24(4):205-215.
 Abstract
Abstract  PDF
PDF